Introduction
Are you confident that the surgical mask available for your use provides appropriate barrier protection for the task you or your colleagues are about to perform? How many types of surgical masks do you have in your facility? Do you know how to differentiate their barrier protection? Does it matter whether you choose an ASTM Level 1 versus an ASTM Level 3 surgical mask for a procedure where high amounts of splash and spray are anticipated? These are a few of the questions you should consider when pondering the question: Is Your Surgical Mask Protecting You? To answer these questions, healthcare personnel should be aware of the relevant standards, guidelines, and professional recommendations that address surgical masks. Through this knowledge, they are better equipped to make appropriate mask selections; thereby reducing exposure to blood, bodily fluids, and other potentially infectious material (OPIM).
Standards and Guidelines Related to Surgical Masks
Several organizations have published standards and guidelines pertaining to the performance, classification, and use of surgical masks. These organizations include the United States Food and Drug Administration (FDA) and ASTM (formerly known as the American Society for Testing Materials) International. Guidelines related to the use and selection of surgical masks include those published by the Association of periOperative Registered Nurses (AORN). Those guidelines will be addressed in a later section.
Food and Drug Administration (FDA)
The FDA is responsible for protecting the public health by assuring the safety, effectiveness, quality, and security of medical devices. The FDA has identified surgical masks as surgical apparel under the surgical devices category. Surgical masks have been given a Class II classification and, prior to introduction into interstate commerce, are subject to premarket notification where manufacturers submit a Premarket Notification (510(k)) to the FDA. For premarket notification [510(k)] of surgical masks, the FDA recommends that five material performance characteristics be tested and reported. These performance characteristics are fluid resistance, differential pressure, bacterial filtration efficiency (BFE), particulate filtration efficiency (PFE), and flame spread.1 Tests performed as specified by the ASTM F2100-11 Standard evaluate these performance characteristics. The following is a review of this standard and the related testing methods.
ASTM F2100-11: Standard Specification for Performance of Materials Used in Medical Face Masks.2 For face masks, ASTM F2100-11 provides classifications of medical face mask material performance. It is important to note that this Standard does NOT evaluate face masks for regulatory approval as respirators, nor does it address all aspects of face mask design and performance. For instance, it does not evaluate the effectiveness of face mask designs as related to barrier and breathability properties.
The ASTM F2100-11 standard specifies five tests that evaluate the material’s fluid resistance, differential pressure (breathability), BFE, PFE, and flame spread. While the five required tests remain the same from earlier versions of this standard, the classifications were changed from “Performance Class” (Low, Moderate, and High) to “Performance Level” (1, 2, and 3). And, importantly, a graphic of the Performance Level is required on the labeling of the primary mask packaging.
Testing Methods
The five tested face mask characteristics and test methods specified in the ASTM F2100-11 Standard are noted in Table 1.
Related Article
If the tested material has passed the test criteria, it may be identified as an ASTM Level 1, 2, or 3 level mask, depending on the test results. The following is an overview of these test methods.
- ASTM F1862 Standard Test Method for Resistance of Medical Face Masks to Penetration by Synthetic Blood (Horizontal Projection of Fixed Volume at a Known Velocity).3 This test allows face mask materials to be ranked according to their ability to resist synthetic fluid penetration. For this test, face masks are challenged with synthetic blood at various levels of pressure (80, 120, 160 mm). The higher the pressure withstood, the greater the fluid spray and splash resistance.
- Differential Pressure (Delta-P).1 This is a clinical test that measures and reports mask breathability. The differential pressure is the measured pressure drop across a surgical face mask material. It determines the resistance of a surgical face mask to air flowing through it; the pressure drop also relates to the breathability (i.e., how easily air passes through the mask) as well as the comfort of the surgical mask.
Industry standards measure breathability from the most to the least. In general, the lower the Delta-P score, the more breathable the mask is. For example, a mask with a Delta-P score of 1 to 2 is often described by the wearer as comfortable and very cool, while a mask with a Delta-P score of 5 or above may be considered hot and uncomfortable. Typically, a fluid resistant mask will have a mid-range score; whereas a respirator will have a differential pressure of 4, 5, or higher, and a mask with no fluid resistance will have a lower score on the comfort scale.
- ASTM F2101: Standard Test Method for Evaluating the Bacterial Filtration Efficiency (BFE) of Medical Face Mask Materials, Using a Biological Aerosol of Staphylococcus aureus.4 An important measure of a material’s ability to provide an adequate barrier to the transfer of microorganisms is its BFE (i.e., the ability of a material to prevent the passage of aerosolized bacteria). The ASTM F2101 is the standard test method used to measure the BFE of medical face mask materials as an item of protective clothing; although not required, it can also be performed on other barrier fabrics.
The ASTM F2101 test is used to determine the amount of infective agent that is retained by the surgical face mask, which is directly related to the amount of bacteria released through the mask into the air. It is important to note that this test method does not define acceptable levels of BFE, but establishes a basis for comparison of different medical face mask materials. Therefore, when looking at the results for this test method, it is necessary to understand the specific condition under which testing is conducted. The maximum BFE that can be determined by this method is 99.9%. A higher BFE percentage indicates a better protection level for the patient against infective agents from the healthcare staff and better protection for the wearer.
To put this in perspective, Table 2 highlights a hypothetical situation where barrier fabrics with BFE percentages ranging from 50 to 99 are challenged with 2,200 bacteria. Note in the case of fabric with the BFE of 50%, 1,100 or half of the bacteria penetrate. While, in the case of fabric with 99% BFE, only 22 bacteria penetrate the fabric.
- ASTM F2299 Standard Test Method for Determining the Initial Efficiency of Materials Used in Medical Face Masks to Penetration by Particulates Using Latex Sphere.5
This test method measures the submicron particulate filtration efficiency (PFE) and the effectiveness of a material to filter aerosolized particles. It measures the initial filtration efficiency of materials used in medical face masks by sampling representative volumes of the upstream and downstream latex aerosol concentrations in a controlled airflow chamber. Specific test techniques are provided for both manufacturers and users to evaluate materials when exposed to aerosol particle sizes between 0.1 and 5.0 μm. As with the BFE test, the results are expressed in the percentage that does not pass through the fabric at a given aerosol flow rate. - 16 CFR (Code of Federal Regulations) Part 1610: Standard for the Flammability of Clothing Textiles. The material to be tested is held in a special apparatus at an angle of 45°. A standardized flame is applied to the surface near the lower end of the material for 1 second. When tested as described in section 1610.6, the material is classified as Class 1, Normal flammability, when the burn time is 3.5 seconds or more. 6,7 The fifth test evaluates the flammability of the mask material.
Not All Surgical Masks are Created Equal
The term surgical mask refers to an FDA-cleared laser, isolation, or medical procedure mask, with or without a face shield, worn to protect surgical patients and healthcare team members from transfer of microorganisms and body fluids. Surgical masks are intended to protect the wearer’s face from large droplets (>5 micrometer in size) & splashes of blood and other body fluids. Surgical and high-filtration surgical laser masks DO NOT provide the degree of protection to be considered respiratory personal protective equipment (PPE).8
There are differences in surgical mask performance levels, construction, and fit.
Levels of Performance for Surgical Masks
As summarized in Table 3 to achieve a Level 1 rating, the mask material must pass the fluid resistance test at 80 mm Hg, a differential pressure with less than 4 mm H2O, a BFE and PFE at 95% or greater, and a Class 1 flame spread. To achieve a Level 2 rating, the mask material must pass the fluid resistance test at 120 mm Hg, a differential pressure with less than 5 mm H2O , the BFE and PFE at 98% or greater, and a Class 1 flame spread. To achieve a Level 3 – THE HIGHEST rating – the only test result that has changed from the Level 2 requirements is the fluid resistance. A mask material that is rated at level 3 must pass the fluid resistance challenge at 160 mm Hg or higher.2
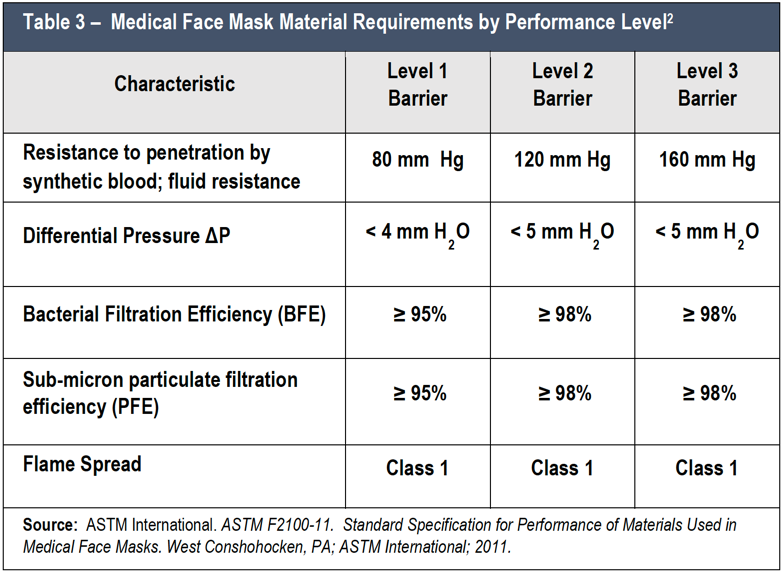
Studies have shown that the filtration efficiency for surgical masks is highly variable depending on the type of mask and manufacturer.8,9 Therefore, it is important for end users to check the mask and mask packaging for information on factors that include whether the mask is FDA cleared and whether it has achieved a performance level (i.e., level 1, level 2, or level 3) as specified by the ASTM F2100-11 Standard.
Construction and Fit of Surgical Masks
Masks vary in design features (e.g., tie-on, ear-loop, attached face shield) and style (e.g., duck bill, flat pleated, cone shaped, pouch). Material composition (i.e., type of fabric, metals, and elastic materials) as well as size and dimensions vary as well.1 As noted by OSHA, surgical masks are not designed or certified to prevent the inhalation of small airborne contaminants. Nor are they designed to seal tightly against the user’s face. During inhalation, much of the potentially contaminated air can pass through gaps between the face and the surgical mask and not be pulled through the filter material of the mask. Their ability to filter small particles varies significantly based upon the type of material used to make the surgical mask. Only surgical masks that are cleared by the U.S. food and Drug Administration to be legally marketed in the United States have been tested for their ability to resist blood and body fluids.9
The fit of a face mask is fundamental to overall performance and barrier protection. As examples, a face mask that is too small or too big will fail to provide the wearer with an adequate seal around the nose and mouth. It is important to note that even when surgical masks with the most efficient filter media (i.e., high-filtration surgical laser masks) are properly worn, the fit factor obtained is quite low.8,10 This highlights the importance of appropriate mask size and best facial fit to maximize the benefits of wearing a surgical mask. In addition, healthcare personnel should refer to their professional organization(s) for guidance. As an example, on their Clinical FAQ website, AORN has posted the following guidance on masks with ear loops. “Masks with ear loops may not … provide a secure facial fit that prevents venting at the sides of the mask. The surgical mask should cover the mouth and nose and be secured in a manner that prevents venting at the sides of the mask. A mask that conforms to the perioperative team member’s face decreases the risk of the perioperative team member transmitting nasopharyngeal and respiratory microorganisms to the patient or the sterile field.”11
Strategies for Appropriate Selection of Surgical Masks
Strategies for appropriate selection of surgical masks include using professional guidelines and resources. Recommendations related to surgical masks include guidance from the Health Care Infection Control Practices Advisory Committee (i.e., the federal advisory committee that provides advice and guidance to the Centers for Disease Control and Prevention)12 and the Occupational Safety and Health Administration (OSHA). The OSHA mandate states that when health care workers are performing activities that generate splashes, sprays, aerosols of blood or OPIM, they must wear appropriate facial protection to protect them from exposure. 13
As outlined below, additional guidance for selection and use of surgical masks can be obtained from the Association of periOperative Registered Nurses (AORN) guidelines and applying knowledge of the ASTM F2100-11 Standard Specification for Performance of Materials Used in Medical Face Masks.
Association of periOperative Registered Nurses (AORN)
This professional association provides and regularly updates several recommendations related to the performance characteristics and comfort of surgical PPE that can be found in the AORN guidelines as summarized below.
Guideline for Product Selection14
This guideline provides recommendations for evaluating and purchasing medical devices and other products used for patient care including the following.
A mechanism to select products should be developed; this includes establishing a multidisciplinary product evaluation and selection committee.
- The multidisciplinary committee should develop a process that guides product selection.
- Consistent requirements for each product being evaluated should be determined. Examples of product-specific requirements include:
- compatibility with existing methods of disposal and reprocessing
- procedure-related requirements (e.g., resistance to penetration by blood and other body fluids)
- end-user preferences and requirements (e.g., for a surgical gown: comfort, the amount of protection from blood and body fluids, size, free from toxic ingredients or allergens)
- patient-related requirements (e.g., presence of infectious diseases)
-
compliance with federal, state, local regulatory agencies, and with standard-setting bodies
- A product-specific evaluation tool should be developed with the use of unique, product-specific criteria, including:
- safety, performance, quality
- efficiency, ease of use
- effect on quality patient care and clinical outcomes
- evidence-based efficacy
- analysis of the financial impact
- sterilization/reprocessing parameters (including degree of difficulty)
- environmental impact
- quality of the manufacturer’s instructions
Guidelines for Prevention of Transmissible Infections 15,16
These guidelines provide guidance in implementing standard and transmission-based precautions (i.e., contact, droplet, and airborne precautions) to reduce the risk for infection. Further guidance is as follows.
- Masks provide protection for the wearer’s mucous membranes of the nose and mouth, which are susceptible to infections pathogens.
- Masks are also used as a component of standard and droplet precautions to prevent contact with respiratory secretions or blood and body fluid sprays.
- Surgical masks that are tested and evaluated for fluid resistance, bacterial filtration efficiency, differential pressure, as well as flammability, are appropriate components of PPE in the perioperative practice setting.
Guideline for Surgical Attire17,18,19
According to the AORN Guideline for Surgical Attire, all personnel who enter the restricted areas should wear a surgical mask when open sterile supplies and equipment are present. A surgical mask should be worn to protect both the healthcare team member as well as the patient from the transfer of microorganisms. The surgical mask protects health care workers from droplets that are larger than 5 micrometers in size. A single surgical mask is worn to protect the health care worker from contact with infectious material from the patient (eg, sprays or spatters of blood or body fluids, respiratory secretions) and to protect the patient from exposure to infectious microorganisms carried in the health care worker’s mouth or nose. A surgical mask protects the nose and mouth of personnel from inadvertent splashes or splatters of blood and other body fluids. Other best practices that improve barrier effectiveness and reduce the potential for contamination of surgical masks include the following.
The mask should completely cover the nose and mouth.
- The mask should be secured in a manner that prevents venting (i.e., securely tied in the back of the headand also behind the neck). Infectious agents can reach the wearer’s nose and mouth if leaks at the face-mask seal are present.
- A fresh, clean surgical mask should be worn for every procedure.
- A mask that becomes wet or soiled should be replaced. When a mask becomes wet, its filtering capacityis compromised; the microbial barrier efficacy of surgical masks with a bacterial filtration efficiency of95% decreases after four hours.
- Masks should not be worn dangling or hanging down from the neck.
- Masks should be discarded after each procedure.
- The mask should be removed carefully by handling only the mask ties.
- Hand hygiene should be performed after the mask is removed.
Guideline for Energy Devices20
In regards to protecting perioperative personnel from surgical smoke generated during surgical procedures, this AORN Guideline recommends respiratory protection (e.g., a fit-tested surgical N95 respirator) as a secondary protection against residual surgical smoke. It is important to note that general room ventilation and smoke evacuation (i.e., local exhaust ventilation) are the first lines of protection against surgical smoke inhalation, not high-filtration masks.
It should also be noted that both surgical and high-filtration surgical laser masks do not provide enough protection to be considered respiratory PPE.8 Therefore, the potential for exposure to airborne contaminants and infectious agents, including those in surgical smoke21, dictates the use of respiratory PPE, such as a surgical N95 particulate respirator. These respirators are designed to protect the user from both droplet and airborne particles and greatly decrease a large size range of particles from entering the wearer’s breathing zone. All health care workers who must use a respirator to control exposure to hazardous agents in the workplace must be trained on the proper use of the respirator and also pass a fit test before it can be used in the workplace.
ASTM F2100-11 Standard Specification for Performance of Materials Used in Medical Face Masks
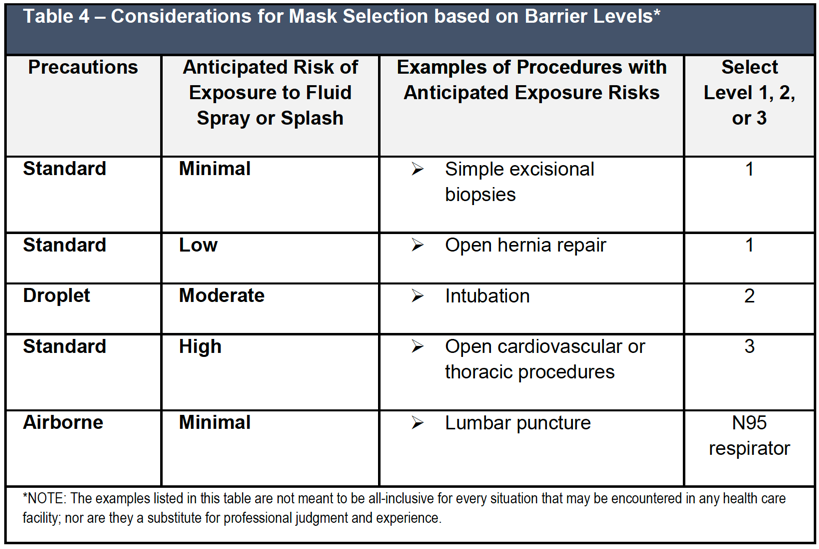
Healthcare personnel should use their knowledge from ASTM F2100-11 when selecting masks, giving
consideration to whether the patient is under standard precautions or extended precautions, the type of
procedure to be performed, and the anticipated amount of splash or spray.
Table 4 outlines examples of considerations in mask selection based on barrier levels. Note, this table is intended as a tool to provide general guidelines as a starting point for decision-making and is not a substitute for professional judgment and experience.
Summary
During invasive procedures, both the patient and members of the health care team are at risk for exposure to infectious agents through pathogen penetration of surgical masks. Therefore, selection of surgical masks with appropriate barrier protection for the task at hand is essential. This selection process is challenging as surgical masks differ in barrier performance levels, construction and fit. Organizations that have published standards and guidelines pertaining to the performance, classification, selection, and use of surgical masks include the FDA, ASTM International, and AORN. Health carepersonnel should be knowledgeable of information provided by these organizations and use this knowledge to enable them to make appropriate surgical mask selections.
Sources
-
Food and Drug Administration. Guidance for industry and FDA staff: surgical masks – premarket notification [510(k)] submissions; guidance for industry and FDA. Document issued on March 5, 2004 and a correction posted on July 14, 2004. . Accessed 6.14.17.
-
ASTM International. ASTM F2100 – 11. Standard Specification for Performance of Materials Used in Medical Face Masks West Conshohocken, PA; ASTM International; 2011.
-
ASTM International. ASTM F1862M-13. Standard Test Method for Resistance of Medical Face Masks to Penetration by Synthetic Blood (Horizontal Projection of Fixed Volume at a Known Velocity). West Conshohocken, PA; ASTM International; 2013.
-
ASTM International. ASTM F2101-07. Standard Test Method for Evaluating the Bacterial Filtration
Efficiency (BFE) of Medical Face Mask Materials, Using a Biological Aerosol of Staphylococcus aureus.
West Conshohocken, PA; ASTM International; 2007.
-
ASTM F2299 / F2299M-03(2010), Standard Test Method for Determining the Initial Efficiency of Materials Used in Medical Face Masks to Penetration by Particulates Using Latex Spheres, ASTM International, West Conshohocken, PA, 2010.
-
Federal Register Consumer Product Safety Commission. CFR Part 1610: Standard for the flammability of clothing textiles; proposed rule. Part II Consumer Product Safety Commission. Tuesday, February 27, 2007. https://www.cpsc.gov/PageFiles/95108/clothingflammstd.pdf. Accessed June 14, 2017.
-
Federal Register Consumer Product Safety Commission. CFR Part 1610.6. Requirements for classifying textiles. In 16 CFR Part 1610 Standard for the flammability of clothing textiles; proposed rule. https://www.law.cornell.edu/cfr/text/16/1610.6. Accessed June 14, 2017.
-
Benson SM, Novak DA, Ogg MJ. Proper use of surgical N95 respirators and surgical masks in the OR. AORN J. 2013;97(4):458-467.
-
OSHA. (2009). OSHA FactSheet – Respiratory Infection Control: Respirators Versus Surgical Masks. Accessed June 14, 2017.
-
Oberg, T. and L. M. Brosseau (2008). “Surgical mask filter and fit performance.” Am J Infect Control 36(4): 276-282.
-
Association of periOperative Registered Nurses. November 6, 2014. Are Masks with Ear Loops Acceptable in the periOperative Environment? Clinical FAQs: Surgical Attire. Accessed June 17, 2017.
-
Siegel, J. D., E. Rhinehart, et al. 2007. Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Health Care Settings. Am J Infect Control 35(10 Suppl 2): S65-164.
-
OSHA. Occupational Safety and Health Standards, Toxic and Hazardous Substances, Bloodborne Pathogens, United States Department of Labor. 1910.1030. https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=10051. Accessed June 3, 2017.
-
Association of periOperative Registered Nurses. 2017. Guideline for Product Selection. AORN Guidelines for Perioperative Practice, AORN, Inc.: 183-190.
-
Association of periOperative Registered Nurses. 2017. Ambulatory Supplement: Guideline for Prevention of Transmissible Infections. AORN Guidelines for Perioperative Practice, AORN, Inc.: 540-542.
-
Association of periOperative Registered Nurses. 2017. Guideline for Prevention of Transmissible Infections. AORN Guidelines for Perioperative Practice, AORN, Inc.: 507-539.
-
Association of periOperative Registered Nurses. 2017. Guideline for Surgical Attire. AORN Guidelines for Perioperative Practice. AORN, Inc.; 105-127.
-
Braswell, M. L. and L. Spruce (2012). “Implementing AORN recommended practices for surgical attire.” AORN J 95(1): 122-137; quiz 138-140.
-
Cowperthwaite, L. and R. L. Holm (2015). “Guideline implementation: Surgical attire.” AORN J 101(2): 188-194; quiz 195-187.
-
Association of periOperative Registered Nurses. 2017. Guideline for Safe Use of Energy-Generating Devices. AORN Guidelines for Perioperative Practice, AORN, Inc.: 129-156.
-
Gao, S., R. H. Koehler, et al. (2016). “Performance of Facepiece Respirators and Surgical Masks against Surgical Smoke: Simulated Workplace Protection Factor Study.” Ann Occup Hyg 60(5): 608-618.